Introduction
Gastric dilatation-volvulus (GDV) syndrome is an acute, life-threatening condition requiring immediate emergency surgical intervention.
Intervention includes stabilization, decompression, derotation, and gastropexy. Every second counts!
It is most commonly seen in large to giant breed dogs, particularly in deep-chested dogs, although any breed may be at risk.
It can occur when the stomach fills with gas, food, and/or fluid, resulting in gastric dilatation, or otherwise known as “bloat”. Gastric dilatation can then progress further into a volvulus. Volvulus is when the stomach “flips” or twists upon itself (i.e. gastric rotation). Once a volvulus has occurred, the stomach continues to fill with gas, and thereby continuing to dilate/distend.
Note that the pathophysiology involves two theories: gastric rotation followed by gastric dilatation versus gastric dilatation followed by gastric rotation.
A GDV not only obstructs the stomach’s inflow and outflow, but additionally, the enlarged gas-filled stomach presses dorsally against the caudal vena cava, resulting in cutting off blood circulation throughout the entire body, known as obstructive shock. This obstructive shock leads to decreased cardiac output, reduced blood flow, hypovolemia, and hypotension. Blood flow is further reduced by fluid entrapment within the stomach and third space shifting, leading to hypovolemic shock. Reduced blood flow and oxygen delivery results in compromise to vital tissues and organs. The increased pressure within the stomach can additionally cause stomach wall destruction, tissue necrosis, stomach perforation, and peritonitis. Tissue necrosis and toxin buildup can lead to distributive shock. Immediate emergency surgical intervention is required to help prevent and/or treat life-threatening sequelae such as shock, cardiac arrhythmias, ventilation/perfusion mismatch, organ damage, electrolyte disturbances, acid-base derangements, tissue necrosis, sepsis, and/or disseminated intravascular coagulation. Sequelae are described in more detail below in the “Pathophysiology” section.
Incidence
The incidence of GDV is 2.4 to 7.6 for every 1,000 dogs admitted to a hospital, and it is the second leading cause of canine death with 15% dying due to the condition within their lifetime.
Large to giant breed dogs have a 3.9% to 36.7% lifetime risk.
Risk Factors
Some factors that can increase GDV risk include:
- Ingesting a large amount of food or food particles less than 30 mm in size
- Eating only one meal a day
- Running/exercise after eating a large meal or drinking a large volume of water
- Anxiety or having a nervous temperament (suspected due to increasing the amount of swallowed air)
- Advanced age: risk by a factor of one
- Giant breed status: in urban residences, 22% of cases
- Large breed status: in rural residences, 24% of cases, a risk factor of one
- Having a history of GDV in a first degree relative
- Having a history of a splenic torsion/splenectomy: 3% to 8% incidence
- Presence of a gastric foreign body: by a factor of five, a 98% increase
- Splenectomy: intact male in 25% of cases
- Higher body weights or older age are more likely to have a history of a splenectomy
- GDV without a history of splenectomy: 1% to 6% of cases
- GDV with a history of splenectomy: 3% to 6% of cases, risk factor of 5.3 (GDV develops 350 days post-operatively (PO)) with GDV by 1yr, 3yrs, & 6yrs PO having a 65%, 20%, & 15% incidence respectively, versus no GDV by 1yr, 3yrs, & 6yrs PO having a 50%, 30%, & 20% incidence respectively
Predisposition
Single-nucleotide polymorphism array genotyping found the most significant signal in Collies, German Shorthaired Pointers, and Great Danes. Genes for gastric tone and motility were involved (VHL, NALCN, and PRKCZ).
Higher-risk breeds include (but not limited to):
- Airedale terrier
- Akita
- Basset Hound
- Borzoi
- Bull Mastiff
- Chow Chow
- Collie
- Doberman Pinscher (4% incidence)
- German Shepherd (12% to 25.8% incidence)
- Gordon Setter
- Great Dane (11% incidence, 42% life-time risk)
- Irish Setter
- Irish Wolfhound
- Rottweiler (4% life-time risk)
- Saint Bernard
- Standard Poodle (5% incidence)
- Weimaraner
Males (overall 59.25%, intact 53.1%), older dogs (5 to 10 years of age make up 55% of dogs, dogs 6 to 7 years of age are 2x as likely to be affected compared to those 3 to 4 years of age), and dogs with higher body weights are over-represented.
Pathophysiology
The pathophysiology involves two theories: gastric rotation followed by gastric dilatation versus gastric dilatation followed by gastric rotation.
With either, the stomach rotates about its long axis in a clockwise direction, with the pylorus and proximal duodenum displacing ventral and cranial, from right to left, and finally positioning dorsal to the esophagus.
The gastroesophageal sphincter is obstructed and pyloric dysfunction is evident.
Eructation, vomiting, and pyloric outflow is inhibited, resulting in aerophagia, bacterial fermentation, and rapid gas buildup, and progressive gastric dilatation.
GDV physiologic changes include the following in sequential but also overlapping phases:
- Increased intra-abdominal pressure, decreased venous blood flow, caudal vena cava compression, and obstructive shock
- Poor splanchnic circulation causing small intestinal compromise, increased endotoxin absorption, villi injury, and jejunal mucosal changes
- Decreased pancreatic perfusion, gastric/splenic displacement, and decreased portal venous return; causes pancreatitis and portal hypertension causing decreased hepatic reticuloendothelial function, poor bacterial/endotoxin hepatic clearance, and hepatic ischemia
- Increased gastric volume/abdominal pressure decreases total thoracic volume, increases diaphragmatic pressure and decreased excursions, decreased inspiratory effort, partial lung collapse, ventilation/perfusion mismatch, hypovolemia, decreased pulmonary blood flow, and poor oxygen delivery
- Cardiac dysfunction from poor coronary flow (decreased by 50%), myocardial depressant factor, cardiac arrhythmias (40 to 70% incidence overall, incidence of ventricular arrhythmia 13% to 100%), decreased cardiac output, and increased cardiac oxygen extraction by 30%
- Shock results in poor tissue perfusion and hypoxemia, promotion of anaerobic metabolism with tissue acidosis (hyperlactatemia)
- Full thickness gastric wall necrosis (10% to 37% incidence) from avulsion of the splenic artery and short gastric arteries along the greater curvature (causing a hemoperitoneum), collapsed capillaries (from increased intragastric pressure), and decreased systemic blood pressure (hypotension), causing ischemia, hyperlactatemia, submucosal/mucosal gastric edema, and gastric necrosis
- Other factors include the degree of gastric rotation, duration of GDV, intragastric pressure, gastric wall tension, and systemic arterial pressure
- Splenomegaly from venous congestion/vessel occlusion, intravascular thrombosis, splenic infarction, and splenic necrosis
Ischemic reperfusion injury, sepsis (2.4% incidence), systemic inflammatory response syndrome, and acute respiratory distress syndrome due to reactive oxygen species, directly by lipid peroxidation, and indirectly by neutrophil activation. Poor cardiovascular stability, hypotension, multiple organ failure, and finally death results.
Degree Rotation
Clinical presentations include acute gastric dilation, or gastric dilatation with gastric volvulus.
The former is defined as a 90° counterclockwise gastric rotation around the gastroesophageal junction.
The latter is defined as simultaneous dilation and clockwise 180° to 360° rotation at the gastroesophageal junction. Average rotation reported is between 180° and 270°.
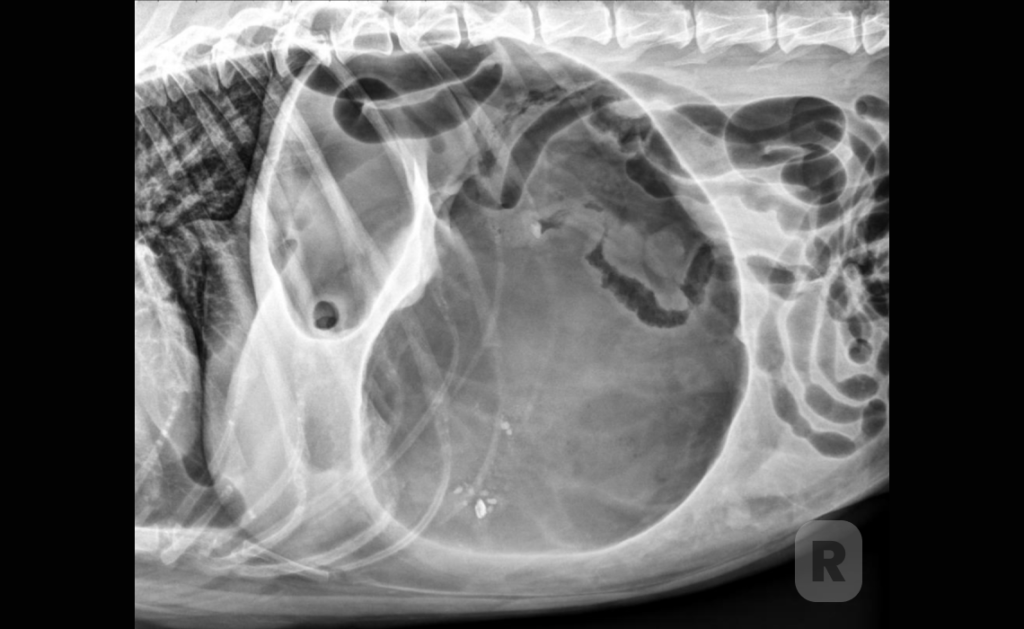
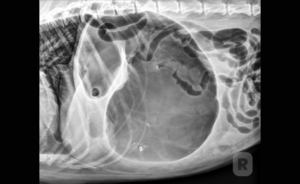
Abdominal Radiography
Findings with the patient in right lateral recumbency include pyloric air entrapment and pyloric
malpositioning that is described as a “double bubble” sign, “reverse C” sign, “boxing glove” sign, “Popeye’s arm” sign, or “Smurf’s hat” sign.
Pneumoperitoneum indicates gastric perforation or gastric trocarization.
Gastric Decompression
Decompression may be via orogastric tube, percutaneous over-the-needle catheter, or trocar.
Delayed Surgery
There is no survival difference for dogs whom received gastric decompression and placement of an indwelling nasogastric tube for 90 minutes prior to surgery versus those with surgery after 5 to 13.7 hours of stabilization.
Of dogs that had surgery in the same anesthetic session as for decompression/stabilization, 33% had gastric necrosis.
Of dogs with surgery at a second anesthetic episode at a mean of 22.3 hours after presentation, 5.7% had gastric necrosis.
Surgical Management
Surgical intervention creates a permanent adhesion between the gastric pyloric antral wall and the right abdominal body wall.
The stomach is retracted to the right abdominal wall and the body of the stomach is pushed dorsally for evaluation of the gastroesophageal junction.
Gastric viability is determined via color, thickness, integrity, and near-infrared fluorescence.
The ideal gastropexy would restore normal anatomic positioning, maintain normal gastric function, is permanent, predictable, simple with minimal complications, and requiring minimal post-operative management.
Surgical Approaches
Surgical approches include:
- Total laparotomy: strength 24.04 N to 106.5 N
- Mini-laparotomy
- Grid approach
- Total laparoscopic: intracorpeally stapled or sutured gastropexy
- Laparoscopic-assisted: Hasson or Veress needle techniques. Strength 45 to 100 N at 30 to 50 days, 51.1 N at 10 weeks.
- Modified laparoscopic-assisted: Strength 35.86 N
- Endoscopic-assisted
Gastropexy Types
Gastropexy types (with associated strengths to the abdominal wall) include:
- Belt loop: seromuscular flap created along the greater curvature of the stomach (gastroepiploic artery), extending to the antrum. A tunnel is created in the abdominal wall, parallel to the gastric incision. Strength 109 N at 50 days
- Modified belt loop
- Circumcostal: single- or double-hinged seromuscular flap is created over the lesser curvature of the stomach or the antrum, by undermining below the muscularis layer. Strength 109 N at 21 days.
- Gastrocolopexy: attaches the greater curvature of the stomach to the transverse colon
- Incisional: most common. Strength 35 to 38 N.
- 59 to 60 N at 3 weeks
- 62 to 106 N at 4 weeks
- 60 to 85 N at 21 to 58 days
- 53 N at 8 weeks
- Modified incisional
- Incorporating: incorporates the line alba laparotomy closure with the gastropexy. Strength 60 to 110 N.
- Muscular flap
- Tube
Splenectomy (15% to 20% need it) and/or gastric resection/invagination (up to 20% need it) may be considered on a case by case basis.
Board Certified Surgeon vs General Practitioner
Deciding to opt for a board certified surgeon versus general practitioner (GP) can be challenging.
The survival rates of patients operated on by GP versus specialists were 81.7% and 88.7%, respectively.
Intraoperative mortality rates for GP versus specialists were 7.0% and 2.9%, respectively. The odds of mortality in dogs operated on by GP were approximately 2x those operated by specialists.
Post-Operative Recovery and Possible Complications
As with other surgical procedures, GDV procedures come with possible complications, including:
- Cardiac arrhythmias: 30% to 50% incidence
- Hemostatic derangements: 20%
- Recurrence of disease: 4% to 10.6%
- Gastrocolopexy: 15%
- Circumcostal: 0% to 7%
- Incorporating: 6.5%
- Incisional: 5%
- Belt loop: 0%
- Coagulopathy: 8%
- Acute renal injury: 3% to 8%
- Peritonitis: 5% (gastric necrosis)
- Splenic thrombosis: 3%
- Pancreatitis: 1.5%
Mortality and Prognostic Factors
Some mortality and prognostic factors include:
- Overall mortality: 10% to 68% (50% due to gastric necrosis)
- Overall survival: 75% to 97%
- No treatment/medical therapy alone: 81% die within one year
- Recurrence rate without gastropexy: 56% within 3 months, 80% long-term
- Splenectomy mortality: 32%
- Partial gastrectomy and splenectomy mortality: 55%
Some high mortality factors include:
- Clinical signs duration > 5 hours: 46% mortality rate
- Depressed/coma at admission: 3x to 36x risk of death
- Coagulopathy: associated with gastric necrosis
- Pre-operative cardiomegaly: survival decreases by 10x
- Survival without gastric resection: 100%, mortality without gastric necrosis: 2% to 14%
- Gastric necrosis: 11x increased mortality
- Gastric necrosis/partial gastrectomy mortality: 31% to 98%, 6x increase in mortality
Prevention
The GDV risk with at-risk patients decreases from 4% to 54.5% to 0.3% to 8% with prophylactic gastropexy.
Mortality decreases by 2x (Rottweilers) to 29.6x (Great Danes) with prophylactic gastropexy.
Gastric motility does not change after prophylactic laparoscopic gastropexy.
Preoperative euthanasia (non-survival without intent to treat) is the majority of GDV mortality. Disease prevention will improve disease mortality greater than improving treatment.
The majority of non-insured dogs (37% to 63%) are euthanized pre-operatively (5 to 7.4x risk), while 0 to 10% of insured dogs are euthanized pre-operatively.
References
- Ignazio S Piras, et al. Identification of Genetic Susceptibility Factors Associated with Canine
Gastric Dilatation-Volvulus. Genes (Basel). 2020 Nov 5;11(11):1313. doi: 10.3390/
genes11111313. PMID: 33167491 PMCID: PMC7694454 DOI: 10.3390/genes11111313. - Aslanian, et al. Gastric dilatation and volvulus in a brachycephalic dog with hiatal hernia.
JAAHA. 2014. May 2014. - Battisi, et al. Gastric foreign body as a risk for gastric dilatation and volvulus in dogs. JAVMA.
- 1190-3.
- Grange, et al. Evaluation of splenectomy as a risk factor for gastric dilatation and volvulus.
JAVMA. 2012. 241. 461-6. - Pipan, et al. Internet based survey of risk factors for gastric dilatation and volvulus dogs and
surgery. JAVMA. 2012. 240. 1456-62. - Sartos, et al. Association between previous splenectomy and gastric dilatation and volvulus in
453 dogs. JAVMA. 2013. - Matteo Olimpo, et al. Gastric Dilatation-Volvulus in Dogs: Analysis of 130 Cases in a Single
Institution. Animals (Basel). 2025 Feb 18;15(4):579. doi: 10.3390/ani15040579. PMID:
40003061 PMCID: PMC11851494 DOI: 10.3390/ani15040579. - Meike Hammer, Jean-Guillaume Grand. Gastric dilatation and volvulus in a 5-month-old
Bernese mountain dog. Can Vet J. 2019 Jun;60(6):587-590. PMID: 31156256 PMCID:
PMC6510282. - Alison K Gardner, Eric L Schroeder. Pathophysiology of intraabdominal hypertension and
abdominal compartment syndrome and relevance to veterinary critical care. J Vet Emerg Crit Care (San Antonio). 2022 Jan;32(S1):48-56. doi: 10.1111/vec.13125. PMID: 35044059 DOI:10.1111/vec.13125. - Beer, et al. Evaluation of plasma lactate concentration and base excess at the time of
hospital admission as predictors of gastric necrosis and outcome and correlation between those
variables in dogs with gastric dilatation-volvulus: 78 cases (2004-2009). JAVMA. 2013
Jan;242(1):54-8. - Green, et al. Evaluation of initial lactate concentration as a predictor of gastric necrosis and
initial lactate as a predictor of survival in 84 dogs with gastric dilatation and volvulus. JVECC.
21(1). 2011. 36-44. - Green, et al. Pre-operative thoracic radiographic findings in dogs presenting for gastric
dilatation-volvulus (2000-2010): 101 cases. JVECC. 2012 October:22(5):595-600. - Zachner, et al. Association between outcome and changes in lactate concentration during
pre-operative treatment in 64 dogs with gastric dilatation and volvulus; 2002-2008. JAVMA. 236; 892-7. - Pablo Espinosa Mur , et al. Radiographic findings in dogs with 360 degrees gastric dilatation
and volvulus. Vet Radiol Ultrasound. 2025 Jan;66(1):e13445. doi: 10.1111/vru.13445. Epub
2024 Oct 10. PMID: 39388661 PMCID: PMC11617607 DOI: 10.1111/vru.13445 . - Jaymie N Zweig , Whitney D DeGroot . Counterclockwise gastric dilatation and volvulus in a
dog. Can Vet J. 2022 Jul;63(7):711-714. PMID: 35784770 PMCID: PMC9207970 . - Patricia Veronica Paravicini , et al. Gastric Dilatation Organoaxial Volvulus in a Dog. J Am
Anim Hosp Assoc. 2020 Jan/Feb;56(1):42-47. doi: 10.5326/JAAHA-MS-6733. Epub 2019 Nov PMID: 31715111 DOI: 10.5326/JAAHA-MS-6733 . - Antonia Lang, et al. [Diagnostic challenges of a 360° gastric dilatation volvulus in a dog: a
case report]. Tierarztl Prax Ausg K Kleintiere Heimtiere. 2025 Jun;53(3):167-172. doi:
10.1055/a-2589-3449. Epub 2025 Jun 13. PMID: 40513607 DOI: 10.1055/a-2589-3449 . - Goodrich, et al. Assessment of two methods of gastric decompression for the initial
management of gastric dilatation-volvulus. JSAP. 2013;54, 75-79. - W Alexander Fox-Alvarez , et al. Evaluation of a novel technique involving ultrasound-
guided, temporary, percutaneous gastropexy and gastrostomy catheter placement for providing
sustained gastric decompression in dogs with gastric dilatation-volvulus. J Am Vet Med Assoc.
2019 Nov 1;255(9):1027-1034. doi: 10.2460/javma.255.9.1027. PMID: 31617809 DOI:
10.2460/javma.255.9.1027. - Russell S White , et al. Evaluation of a staged technique of immediate decompressive and
delayed surgical treatment for gastric dilatation-volvulus in dogs. J Am Vet Med Assoc. 2021
Jan 1;258(1):72-79. doi: 10.2460/javma.258.1.72. PMID: 33314975 DOI:
10.2460/javma.258.1.72 . - Eloïse Lhuillery , et al. Outcomes of dogs undergoing surgery for gastric dilatation volvulus
after rapid versus prolonged medical stabilization. Vet Surg. 2022 Jul;51(5):843-852. doi:
10.1111/vsu.13763. Epub 2022 Jan 6. PMID: 34989433 DOI: 10.1111/vsu.13763 . - Arbough, et al. Biomechanical comparison of Glycomer 631 and Glycomer 631 knotless for
use in canine incisional gastropexy. VetSurg. 2013. - Dujowich, et al. Evaluation of short- and long-term complications post-endoscopic-assisted
gastropexy in dogs. JAVMA. 2010. 236. 177-82. - Dujowich, et al. Evaluation of endoscopically-assisted gastropexy technique in dogs. AJVR.
69 (4): April 2008. - Mathon, et al. A laparoscopic-sutured gastropexy technique in dogs: Mechanical and
functional evaluation. VetSurg. 38: 738-46. 2009. - Mayhew, et al. Prospective evaluation of two intracorporeally sutured prophylactic total
laparopscopic gastropexy technique versus with laparoscopic-assisted gastropexy. VetSurg. 38:
738-46. 2009. - Kaitlyn M Mullen , et al. Use of real-time near-infrared fluorescence to assess gastric viability
in dogs with gastric dilatation volvulus: A case-control study. Vet Surg. 2024 May;53(4):684-694.
doi: 10.1111/vsu.14067. Epub 2023 Dec 22. PMID: 38135927 DOI: 10.1111/vsu.14067 . - Luca Lacitignola , et al. Comparison of total laparoscopic gastropexy with the Ethicon
Securestrap fixation device versus knotless barbed suture in dogs. Vet Rec. 2021
Apr;188(7):e113. doi: 10.1002/vetr.113. Epub 2021 Feb 2. PMID: 33835588 DOI:
10.1002/vetr.113. - Claire Deroy , et al. Simplified Minimally Invasive Surgical Approach for Prophylactic
Laparoscopic Gastropexy in 21 Cases. J Am Anim Hosp Assoc. 2019 May/Jun;55(3):152-159.
doi: 10.5326/JAAHA-MS-6879. Epub 2019 Mar 14. PMID: 30870607 DOI: 10.5326/JAAHA-MS-
6879. - Danielle K Fairfield , et al. Perioperative characteristics and long-term outcomes following
prophylactic total laparoscopic gastropexy using a novel knotless tissue control device in 44
dogs. Can Vet J. 2023 Jul;64(7):659-665. PMID: 37397695 PMCID: PMC10286145. - Dong Woo Kim , et al. Description and biomechanical evaluation of the modified
laparoscopic-assisted percutaneous gastropexy technique in dogs. Front Vet Sci. 2025 Feb
3:11:1509728. doi: 10.3389/fvets.2024.1509728. eCollection 2024. PMID: 39963368 PMCID:
PMC11830685 DOI: 10.3389/fvets.2024.1509728. - Federico Massari , Gary Matthew Martin Kelly . Learning Curve in Two-Port Laparoscopic
Gastropexy Using FlexDex. Animals (Basel). 2024 Jul 9;14(14):2016. doi:
10.3390/ani14142016. PMID: 39061478 PMCID: PMC11274303 DOI: 10.3390/ani14142016. - Luca Formaggini , Matteo Tommasini Degna . A Prospective Evaluation of a Modified Belt-
Loop Gastropexy in 100 Dogs with Gastric Dilatation-Volvulus. J Am Anim Hosp Assoc. 2018
Sep/Oct;54(5):239-245. doi: 10.5326/JAAHA-MS-6596. Epub 2018 Jul 24. PMID: 30040446
DOI: 10.5326/JAAHA-MS-6596 . - F A Mann , Yi Pan , et al. Comparison of incisional gastropexy with and without addition of
two full-thickness stomach to body wall sutures. J Am Vet Med Assoc. 2023 May
30;261(9):1351-1356. doi: 10.2460/javma.22.11.0492. Print 2023 Sep 1. PMID: 37257832 DOI:
10.2460/javma.22.11.0492 . - K K Song , et al. Retrospective analysis of 736 cases of canine gastric dilatation volvulus.
Aust Vet J. 2020 Jun;98(6):232-238. doi: 10.1111/avj.12942. Epub 2020 Apr 6. PMID: 32253749
DOI: 10.1111/avj.12942 . - Bruchin, et al. Evaluation of lidocaine treatment on frequency of cardiac arrhythmias, acute
kidney injury, and hospitalization time in dogs with gastric dilatation volvulus. JVECC. 2012. - J C Alves , et al. Post-surgical photobiomodulation therapy improves outcomes following
elective gastropexy in dogs. Lasers Med Sci. 2024 Aug 8;39(1):211. doi: 10.1007/s10103-024-
04164-2. PMID: 39115705 PMCID: PMC11310231 DOI: 10.1007/s10103-024-04164-2. - Israeli, et al. Pre-operatively serum pepsinogen-A, canine pancreatic lipase
immunoreactivity, and C-reactive protein for prognostic markers in dogs with gastric dilatation
and volvulus. JVIM. 2012. 920-8. - Mackenzie, et al. Retrospective factors for survival post-operatively: 306 dogs. JAAHA.
2010: 46: 97-102. - Desiree Rosselli . Updated Information on Gastric Dilatation and Volvulus and Gastropexy in
Dogs. Vet Clin North Am Small Anim Pract. 2022 Mar;52(2):317-337. doi:
10.1016/j.cvsm.2021.11.004. Epub 2022 Jan 24. PMID: 35082096 DOI:
10.1016/j.cvsm.2021.11.004 . - Benitez, et al. Efficacy of incisional gastropexy for prevention of GDV in dogs. JAAHA 2013;
49:185-189. - Miranda de la Vega , S Christopher Ralphs . Outcomes and complications of prophylactic
incisional gastropexy in 766 dogs (2009-2019). BMC Res Notes. 2023 Oct 31;16(1):300. doi:
10.1186/s13104-023-06595-6. PMID: 37908004 PMCID: PMC10619303 DOI: 10.1186/s13104-
023-06595-6 . - Runge, et al. Evaluation of single port access gastropexy and ovariectomy using articulating
instruments and angled telescopes in dogs. VetSurg 42 (2013) 807-813. - Claire R Sharp , et al. The pattern of mortality in dogs with gastric dilatation and volvulus. J
Vet Emerg Crit Care (San Antonio). 2020 Mar;30(2):232-238. doi: 10.1111/vec.12932. Epub
2020 Feb 19. PMID: 32077192 DOI: 10.1111/vec.12932. - K A Coleman , et al. Evaluation of gastric motility in nine dogs before and after prophylactic
laparoscopic gastropexy: a pilot study. Aust Vet J. 2019 Jul;97(7):225-230. doi:
10.1111/avj.12829. PMID: 31236930 DOI: 10.1111/avj.12829 . - Manuel Boller , et al. The Effect of Pet Insurance on Presurgical Euthanasia of Dogs With
Gastric Dilatation-Volvulus: A Novel Approach to Quantifying Economic Euthanasia in Veterinary
Emergency Medicine. Front Vet Sci. 2020 Dec 8:7:590615. doi: 10.3389/fvets.2020.590615.
eCollection 2020. PMID: 33364255 PMCID: PMC7752994 DOI: 10.3389/fvets.2020.590615 . - S Anderson , et al. Pet health insurance reduces the likelihood of pre-surgical euthanasia of
dogs with gastric dilatation-volvulus in the emergency room of an Australian referral hospital. N
Z Vet J. 2021 Sep;69(5):267-273. doi: 10.1080/00480169.2021.1920512. Epub 2021 May 25.
PMID: 33896404 DOI: 10.1080/00480169.2021.1920512 . - Amy Molitoris , et al. Early career clinicians euthanize more dogs with nontraumatic
hemoabdomen but not gastric dilatation and volvulus than more experienced clinicians.
J Am Vet Med Assoc. 2022 Jul 28;260(12):1514-1517. doi: 10.2460/javma.22.05.0198. PMID:
35905147 DOI: 10.2460/javma.22.05.0198 . - Hannah Donnelly, Karen Dunne. Canine Prophylactic Gastropexy: A Cross-Sectional Study
of Irish Veterinary Professionals’ Attitudes and Current Practices. Vet Med Sci. 2025
Jul;11(4):e70377. doi: 10.1002/vms3.70377. PMID: 40454814 PMCID: PMC12128143 DOI:
10.1002/vms3.70377.
Dr. Shadi Ireifej, DVM, DACVS-SA
Instagram: Dr.Shadi.Ireifej
Facebook: https://www.facebook.com/shadi.ireifej.54
YouTube: @dr.shadiireifej
X: @Shadi_Ireifej
LinkedIn: https://www.linkedin.com/in/shadi-ireifej-2a43561b4/
Website: www.VetTriage.com
VetTriage
Instagram: VetTriage
Facebook: https://www.facebook.com/televeterinarian
YouTube: @vettriage9959
X: VTriage
LinkedIn: https://www.linkedin.com/company/vettriage/posts/?feedView=all
Website: www.VetTriage.com
Author
-

Dr. Shadi Ireifej attended Cornell University where he received his Doctor of Veterinary Medicine degree in 2006. After Cornell, Dr. Ireifej completed his internship at Angel Animal Medical Center and then two one-year small animal surgical internships at Long Island Veterinary Specialists.
Dr. Ireifej then completed a small animal surgery residency at Long Island Veterinary Specialists in 2012, becoming a Diplomat for the American College of Veterinary Surgeons. Since then Dr. Ireifej has operated in numerous multispecialty and emergency veterinary hospitals across the United States, as both a surgeon, hospital owner and Chief Medical Officer.In 2017, Dr. Ireifej created VetTriage and currently serves as their Chief Medical Officer, focusing on clinic and hospital partnership to aid in teletriage and telementoring, promote excellent virtual care as an extension of those facilities, and helping with work-life imbalance for those partnered facilities.
Shadi has been published in scientific and medical journals and enjoys lecturing to a variety of audiences. He is known for being a positive and energetic force, both professionally and personally, sought after to give in-person and virtual talks, attend podcasts, new interviews, radio shows and more.